Microbial Monitoring of Pharmaceutical Grade Water Systems – Common Misconceptions | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

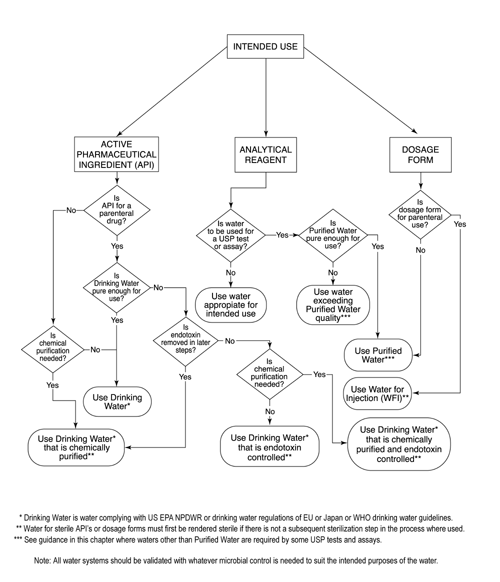
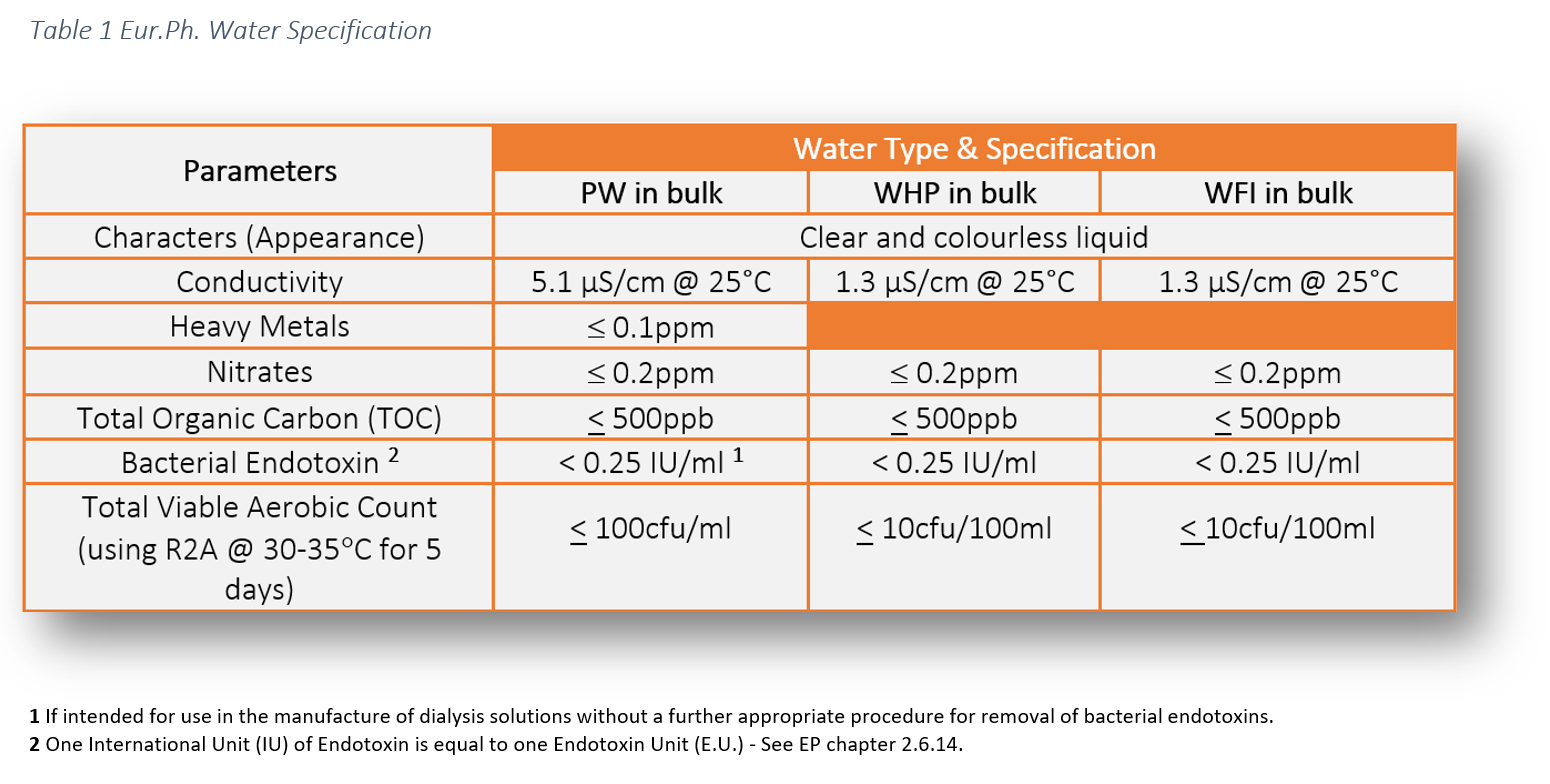
The Design, Control, Monitoring and Validation of Water Systems for Pharmaceuticals, Biologics, Medical Devices, Cosmetics, and Personal Care Products
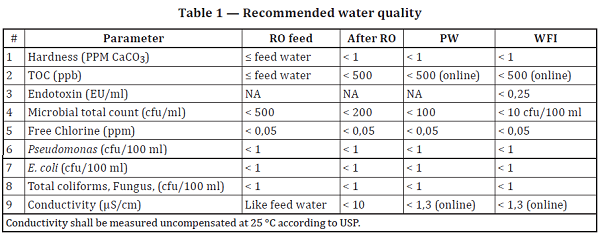
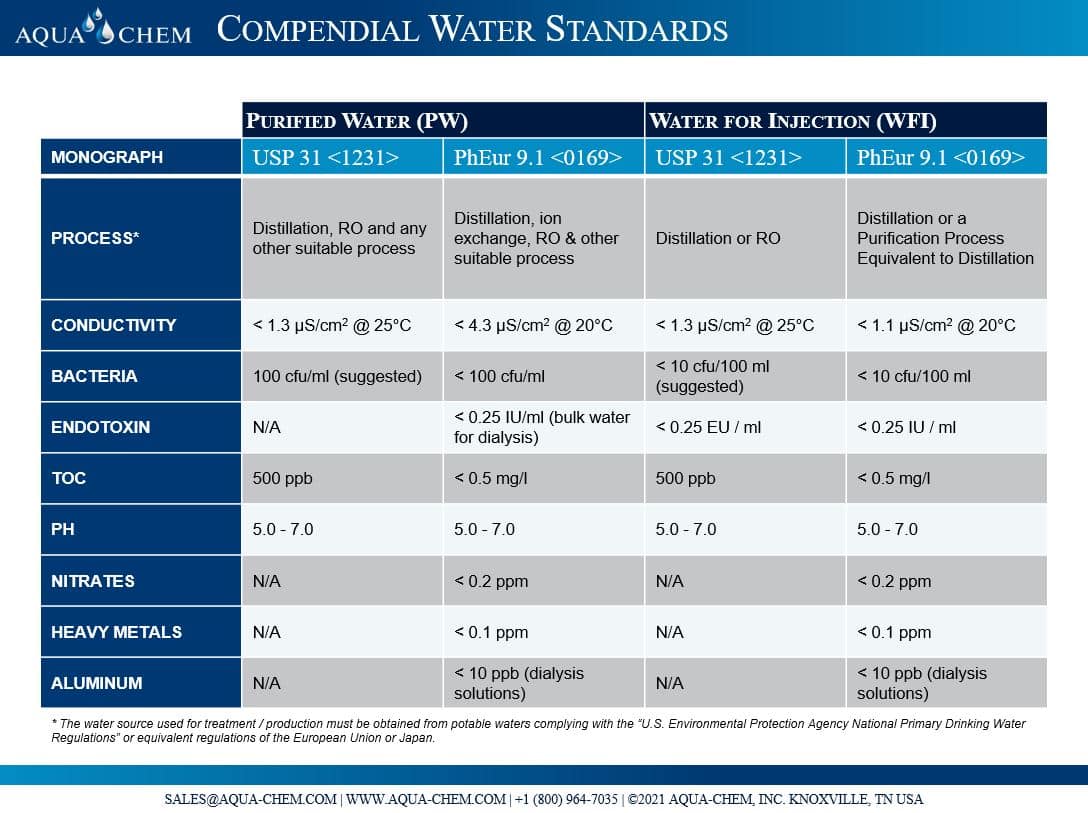
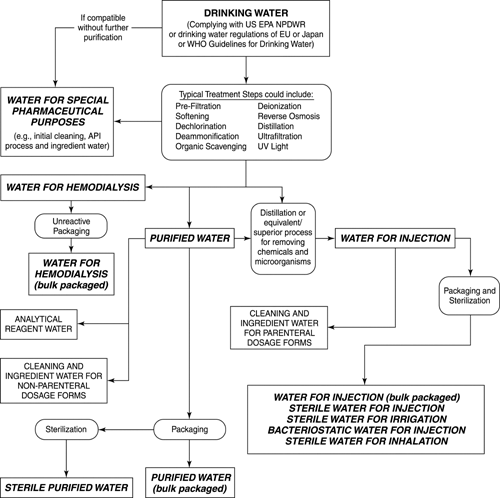
PURIFIED WATER Meets USP/EP Specifications Certificate of Analysis Not for Parenteral use, not for injection, wound irrigation o

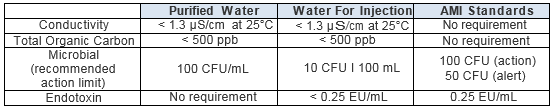
Testing of Purified Water, Raw Materials, In-Process Samples and Finished Non-Sterile Products | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology






![PPT PDF] Pharmaceutical Water System Design Validation – Microbial Testing of Water – Pharmawiki.in PPT PDF] Pharmaceutical Water System Design Validation – Microbial Testing of Water – Pharmawiki.in](https://pharmawiki.in/wp-content/uploads/2017/11/PPT-PDF-Pharmaceutical-Water-System-Design-Validation-Microbial-Testing-of-Water.jpg)